Trulicity: How Do I Use It and What Does It Do?
Medically reviewed by Dr Sultan Linjawi, Endocrinologist & Diabetes Specialist — January 2026

What is Trulicity (Dulaglutide)?
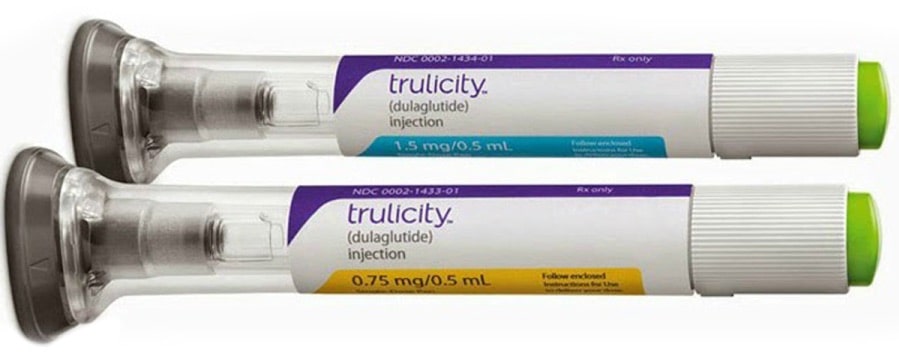
Trulicity is the brand name for a molecule used in the treatment of type 2 diabetes. The generic name is Dulaglutide. It is a relatively new product and is a type of injectable therapy, but it is not insulin. It comes in the form of a disposable single use pen in either 0.75mg dose and 1.5mg dose, making it relatively safe and easy to administer.
Is Trulicity (dulaglutide) the same as insulin?
Trulicity (dulaglutide) is not insulin but is a different type of diabetes medicine called a GLP-1 analogue. It is a small protein just like insulin (called a peptide) and because proteins are easily broken down by stomach acids, which would make the protein inactive, both insulin and Trulicity (dulaglutide) need to be injected under the skin. This allow the molecule to get into the body but bypasses the gut.
What is the dose of Trulicity (Dulaglutide)?
Trulicity is available in either 0.75mg or 1.5mg pens. Generally people should be started on the lower dose and increased after 3 to 6 months. This reduces the side effects substantially. In many countries only 1.5mg is available.
Can higher doses of Trulicity (Dulaglutide) be used?
In the Award 11 Study 1,842 adults with type 2 diabetes were given 1.5mg, 3mg or 4.5mg weekly doses. Those taken the high dose had a greater reduction in Hb A1c (1.53% for the 1.5 mg dose, 1.71% for the 3 mg dose and 1.87% for the 4.5 mg dose. The higher the dose of Trulicity the greater the weight loss with the highest 4.5mg dose associated with about 4.7kg weight loss. The increased doses are now FDA approved in the United States
How often is Trulicity (Dulaglutide) administered?
Trulicity is administered once a week as injection under the skin.
To help remember what day to take your Trulicity, it is useful to have a reminder in your phone each week. It does not matter what day of the week to administer Trulicity (Dulaglutide) so it is best to choose a day that best fits a person's lifestyle. For example, you might choose Sunday as your day to administer Trulicity. You can place a recurring alarm in your phone, so you don't forget. You administer it once every Sunday and that's done. Pretty simple!
Subscribe to Receive One of Our Helpful Free Guides
Click the guide that would be the most useful
Does Trulicity cause weight loss?
Yes. Trulicity can cause weight loss, although it is not a weight loss drug.
There have been a number of studies examining the effectiveness of Trulicity (Dulaglutide) in managing type 2 diabetes. These studies, the AWARD studies, also looked at the associated weight loss in patients using Trulicity. In one of the AWARD studies Efficacy and Safety of Dulaglutide Versus Sitagliptin After 52 Weeks in Type 2 Diabetes in a Randomized Controlled Trial (AWARD-5), it showed that Trulicity was more effective in causing weight loss compared to another type 2 diabetes medication, called sitagliptin (a DPP-4 inhibitor). The average change in body weight was 2.6kg (with the 0.75mg weekly dose of Trulicity) and 3kg (with the 1.5mg weekly dose of Trulicity). This is compared to a 1.53kg weight loss with patients who used sitagliptin.
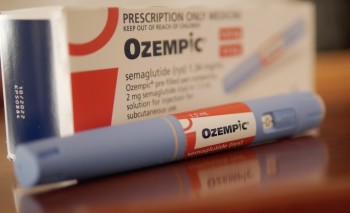
Trulicity (Dulaglutide) vs Ozempic (Semaglutide)
The Sustain 7 study compared the treatment outcomes of Ozempic with Trulicity. Both of these drugs are once weekly GLP-1 analogues used in the treatment of type 2 diabetes.
Ozempic compared to Trulicity led to:
- lower blood sugars based on HbA1c by about 0.3% (-1.4% vs -1.1%).
- Twice as much weight loss with Ozempic (4.6kg-6.5kg depending on dose).
- More people had HbA1c below the 7% to suggest safer sugar levels.
- People using Ozempic had more nausea compared to Trulicity.
Trulicity (Dulaglutide) vs Victoza (Liraglutide)
The AWARD-6 study looked at comparing once weekly Trulicity with once daily Victoza (Liraglutide). The results of this study in people with type 2 diabetes suggests that they are similar in terms of Hb A1c reduction. There was slightly more weight loss with Victoza. Nausea rates were similar with both drugs.
Trulicity (Dulaglutide) alternatives
The following drugs are all based on the GLP-1 or Exendine molecule:
Administered weekly- Ozempic (Semaglutide)
- Trulicity (Dulaglutide)
- Bydureon (Exenatide ER)
Administered daily
- Victoza (Liraglutide)
- Byetta (Exenatide)
- Lyxumia (Lixisenatide)
- Rybelsus (oral Semaglutide)
What are the other effects of Trulicity (Dulaglutide)?
Did you know that GLP-1 has many effects in the body?
As we’ve discussed above, GLP-1 is a hormone that is released from our small intestines in response to food. When GLP-1 is released, it binds to specific GLP-1 receptors. These receptors can be found in various areas of the body (GLP-1 Receptor Localization in Monkey and Human Tissue: Novel Distribution Revealed With Extensively Validated Monoclonal Antibody), including in the:
- pancreas
- stomach
- kidney
- lung
- heart
- skin
- immune cells
- hypothalamus
Please see the following articles for more information:
- What is Ryzodeg insulin and how do I use it?
- Where Do You Keep Your Insulin?
- Managing Diabetes with Insulin: Is it necessary?
- Type 2 diabetes signs and symptoms
- How is Diabetes Diagnosed?
- Prioritising self when you have diabetes – Is it selfish?
Looking for more diabetes videos?
Check out our latest diabetes videos for more great content.
More Articles on Diabetes Treatments
For more information about different Diabetes Treatments, please follow the links below.
Oral Medications for Diabetes
- Starting on Metformin. What you need to know.
- Benefits of Metformin… A Wonder Drug?
- Metformin in Pregnancy. Is it safe?
- Metformin and diarrhoea.
- Vitamin B12 deficiency and diabetes.
- What is an SGLT2 inhibitor? It's a novel way to lower blood sugars, lose weight and maybe live longer.
- Rybelsus (Semaglutide) - A new oral medication to treat Type 2 Diabetes.
---
Insulin for Diabetes
- What is Ryzodeg insulin and how do I use it?
- Fiasp insulin. Fast acting insulin aspart.
- Managing Diabetes with Insulin: Is it necessary?
- Lantas. Solostar insulin glargine.
---
Injections for Diabetes
- What is Trulicity (Dulaglutide) and how does it work?
- Ozempic (Semaglutide) is a new treatment for type 2 diabetes.
- Xultophy 100/3.6 - Type 2 diabetes medication - Benefits, Side Effects and How to Use It.
- What is glucagon and how can a glucagon kit help someone with diabetes?
---
Other Medications for Diabetes
Find the Right Program for You
Select the situation that best describes you
Video Transcript
Trulicity (also known as Dulaglutide) is a treatment for people with Type 2 diabetes. This video explains how Trulicity (Dulaglutide) works, what the side effects are and how to administer it.Today, I'm going to talk about Dulaglutide or Trulicity and specifically I want to show you how to use this pen so you can administer it safely and easily. So this is a new product used widely around the world and it contains a compound called dulaglutide and dulaglutide is essentially a naturally occurring human chemical called GLP-1 or Glucagon-like peptide 1. GLP-1s are in essence chemicals that the body produces in response to food.
It does 3 things, if you eat, it increases insulin production out of the pancreas, which helps lower your blood sugar after eating.
It also interestingly reduces the amount of sugar that the liver is producing. Typically the liver produces sugar, and it does this when you are in periods of starvation. So if you like, it is a storage mechanism that can release sugar when your blood sugars are low and then suck it up when your blood sugars are high. In people with Type 2 diabetes, that process is completely impaired and the liver just produces too much sugar. The third action, which I think is also critical, is it causes a person to feel full. In other words, it increases satiety. So after eating, what you want to do is increase your insulin reduce the liver sugar and feel full.
All of those 3 things are done by GLP-1 and this is a version of GLP-1. One of the clever things about re-packaging GLP-1 and changing slightly the way the molecule is and binding it up to, in this case, an antibody, is that you can change the duration of action.
Trulicity is given once a week. So in order to do it effectively, you choose a day, let's say it is Sunday, something you can remember. You want to put something in your phone so that you get a reminder each week and you don't forget. You administer it once a week, and that's done. Pretty simple.
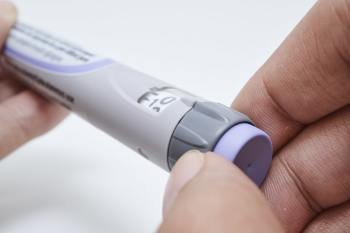
Now let's just look a little bit about how to administer it. On the end of this is a cap, and you would remove the cap. And the needle actually is there but you won't see it because it is hidden within the device, which is quite clever. The other couple of things to say is that there is a lock which is a red triangle and an open and that is so you don't fire the product accidentally when you don't mean to.
So we might look at that in more detail. This is a dummy device, but it does highlight exactly how it gets used. So essentially you take off the cap, you put it against your skin, you move to the unlock position, and press the button. And you know when it's done when you hear the second click. Which will happen any second now. There it is. That is safely and easily disposed of in the bins in the normal way.
But beyond that simple, easy and likely to achieve for a lot of people pretty good sugar control with a bit of weight loss.
Dr Sultan
Frequently Asked Questions
What is Trulicity (Dulaglutide)?
Trulicity is the brand name for dulaglutide, a once-weekly injectable GLP-1 receptor agonist used to treat type 2 diabetes. It is not insulin. Trulicity helps improve blood glucose control and is supplied as single-use prefilled pens in several strengths.
How does Trulicity work?
Trulicity activates the GLP-1 receptor, helping your body lower blood glucose by increasing insulin release when blood sugar is high, reducing how much glucose the liver produces, slowing stomach emptying, and helping you feel fuller sooner.
What is the usual dose of Trulicity?
The typical starting dose is 0.75 mg once weekly. Many people then increase to 1.5 mg each week if more glucose control is needed. In some countries, higher doses (3 mg and 4.5 mg) are available for additional HbA1c and weight benefits.
Can higher doses of Trulicity be used?
Yes. Studies show that doses of 3 mg and 4.5 mg weekly may provide greater improvements in blood glucose and weight. These higher doses are not available in all regions, and suitability depends on individual response and clinical advice.
How do I administer Trulicity?
Trulicity is given as a once-weekly injection using a single-use pen. Remove the cap, place the pen against your skin, unlock it, press the button, and keep it in place until the injection is complete. Dispose of the used pen safely in a sharps container.
Where is the best area of the body to inject Trulicity?
You can inject Trulicity into the abdomen, thigh, or upper arm. If using the same region each week, rotate to a different spot within that area to reduce irritation and ensure consistent absorption.
What should I do if I miss a dose of Trulicity?
If you miss a dose, take it as soon as possible within three days (72 hours). Then continue your usual weekly schedule. If more than three days have passed, skip the missed dose and take your next dose on your normal injection day.
What are the side effects of Trulicity?
Common side effects include nausea, vomiting, diarrhoea, abdominal pain, and reduced appetite, especially when starting or increasing the dose. Less common but serious effects include pancreatitis, allergic reactions, and low blood sugar when used with insulin or sulfonylureas.
Does Trulicity cause thyroid cancer?
Trulicity carries a precaution because high-dose rodent studies showed thyroid tumours, but this has not been demonstrated in humans. It is not recommended for people with a personal or family history of medullary thyroid carcinoma or MEN2.
Does Trulicity cause weight loss?
Many people experience weight loss while using Trulicity. This varies between individuals and is related to reduced appetite and slower stomach emptying. Higher doses often lead to greater weight reduction.
How long does it take for Trulicity to start working?
You may notice improvements in blood glucose within the first few weeks, but full effect is usually seen after 3 to 6 months once the maintenance dose is reached.
How long does Trulicity stay in your system?
Because Trulicity is a once-weekly medicine, it takes several weeks to reach steady state in the body. After stopping treatment, it may take three to five weeks for the medicine to clear completely.